See It in Action
1,300+ research tools connected to FDA, PubMed, Harvard, and more -- all from one dashboard.
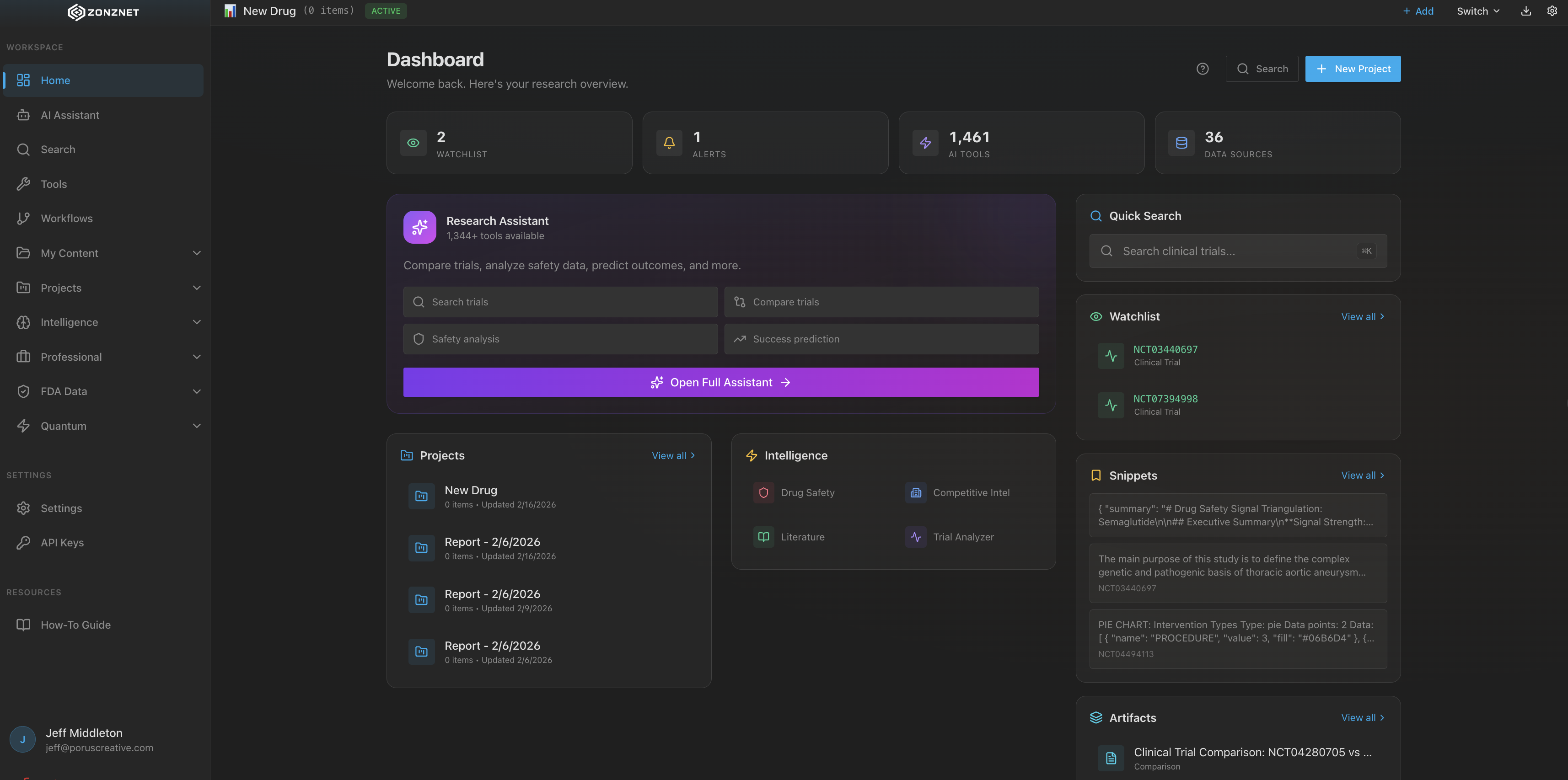
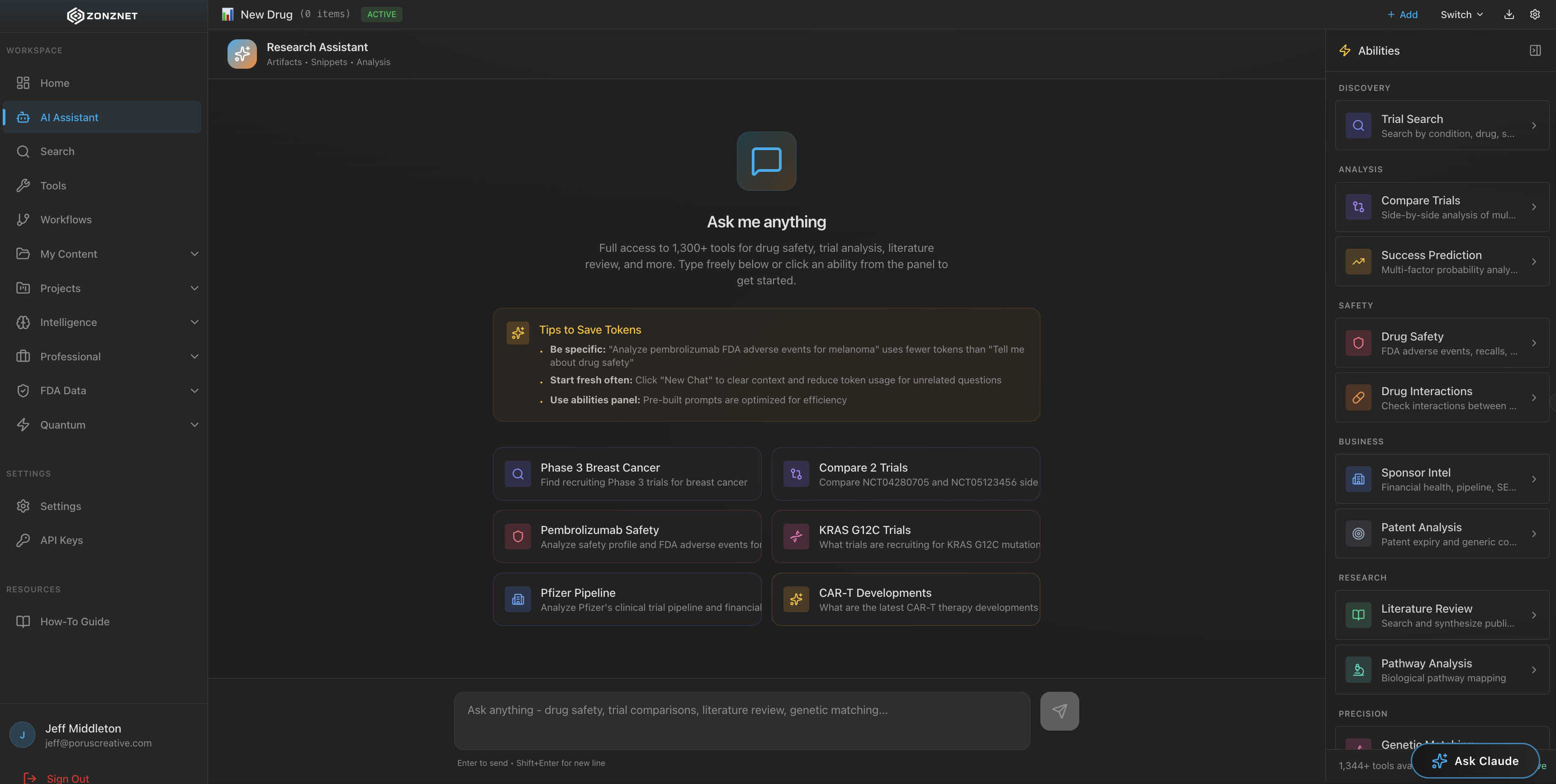
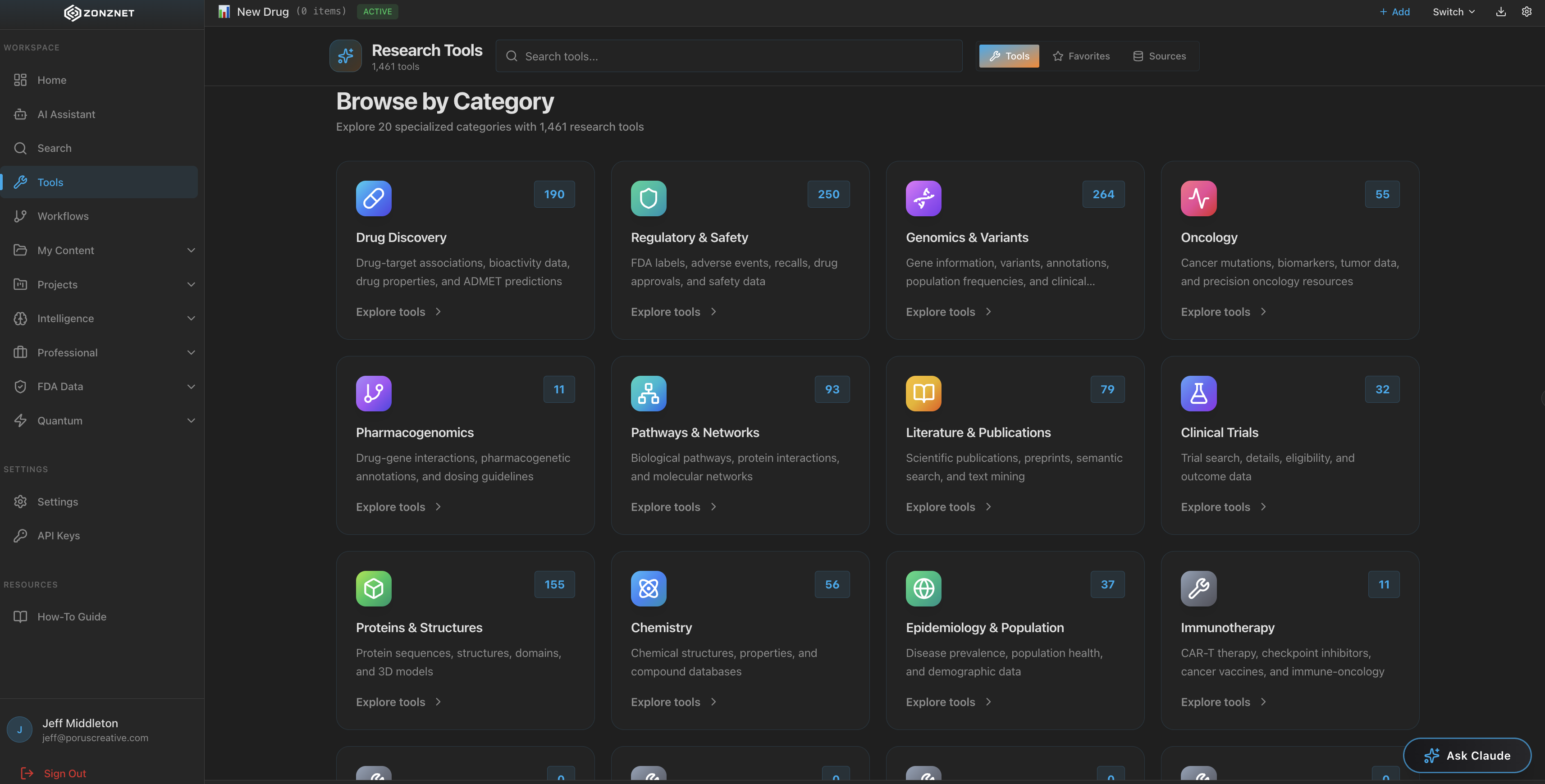
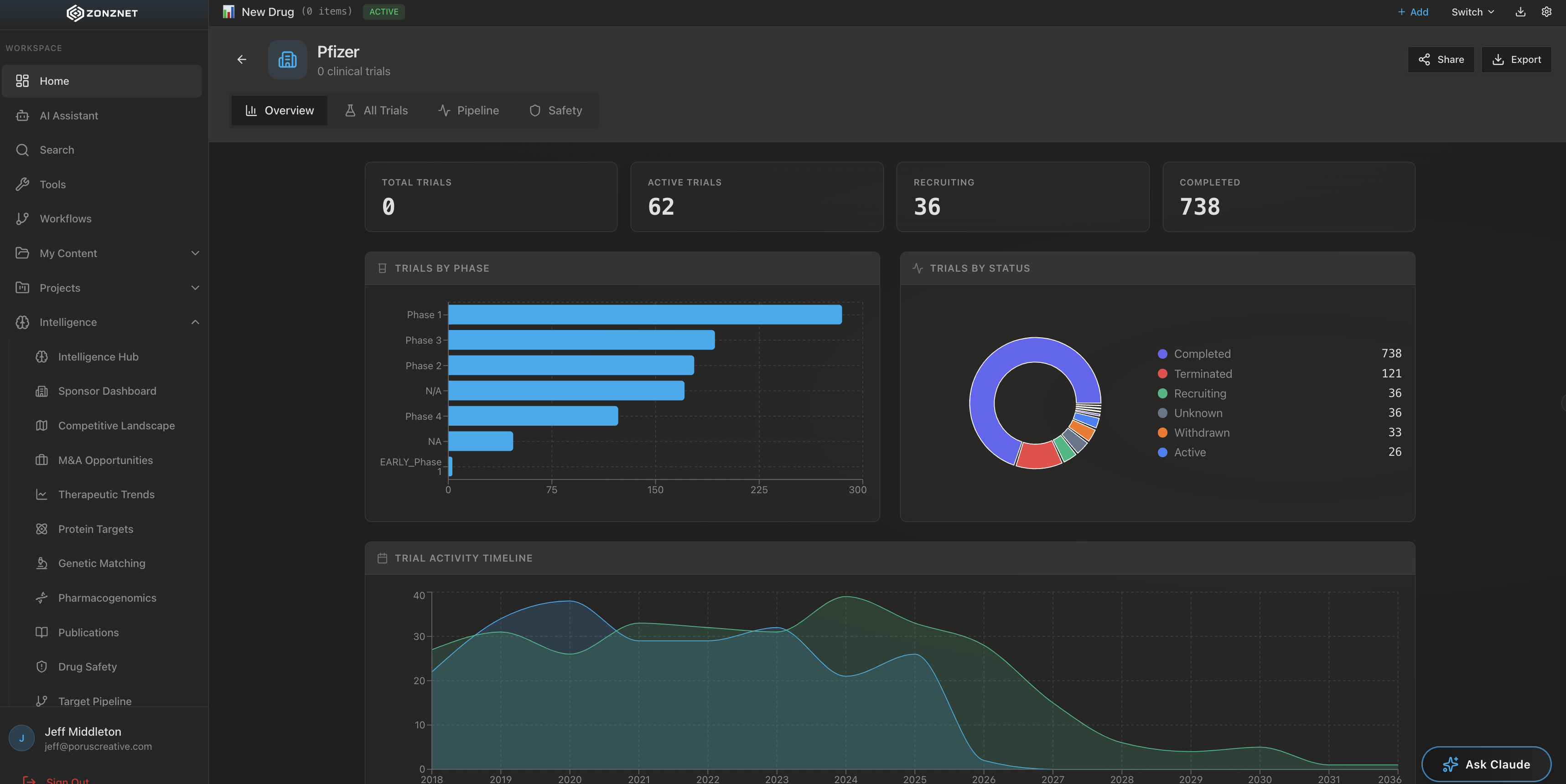
A healthcare drug lookup platform that unifies over 1,300+ tools and connected MCP servers from the FDA, PubMed, Harvard, and dozens of other authoritative medical sources into one powerful search interface.
Look up drug interactions, clinical data, research papers, adverse events, and regulatory information -- all from a single dashboard.
Visit ZonzNet ↗1,300+ research tools connected to FDA, PubMed, Harvard, and more -- all from one dashboard.




Comprehensive healthcare intelligence powered by authoritative medical data sources.
Direct integration with FDA databases including drug approvals, adverse event reports (FAERS), recall data, and labeling information. Access the same data pharmacists and clinicians rely on.
Search millions of peer-reviewed biomedical research articles through PubMed's connected servers. Surface relevant clinical studies, meta-analyses, and systematic reviews in seconds.
Tap into Harvard-affiliated medical research, clinical guidelines, and evidence-based health information. Cross-reference findings from one of the world's most respected medical institutions.
An expansive network of MCP (Model Context Protocol) servers connecting specialized medical tools, databases, and APIs. Each server provides a focused capability -- from drug interaction checking to clinical trial lookups.
One search, many sources. Query across all connected servers simultaneously, with results intelligently aggregated and ranked. No more jumping between ten different medical databases.
Connected servers pull live data from their respective sources, ensuring you always have the most current drug information, safety alerts, and research findings available.
ZonzNet leverages the Model Context Protocol to create a mesh of specialized healthcare data servers, each responsible for a specific domain of medical knowledge.
At its core, ZonzNet connects to over 1,300 MCP servers -- each one a specialized tool with a specific function. Some handle FDA drug label lookups. Others parse PubMed abstracts. Still others cross-reference clinical trial registries. Together, they form a comprehensive healthcare intelligence mesh that no single database could match.
Data quality is paramount in healthcare. ZonzNet prioritizes connections to the most trusted sources in medicine: the U.S. Food and Drug Administration for regulatory data, the National Library of Medicine's PubMed for research, and Harvard-affiliated databases for clinical expertise. Every result is traceable back to its origin.
When you search for a drug, ZonzNet does not just return a list of links. It intelligently queries the relevant MCP servers in parallel, aggregates the results, and presents a unified view that combines regulatory status, clinical research, adverse events, and interaction data into a cohesive, actionable report.
The ZonzNet dashboard is designed for speed and clarity. A single search bar sits at the center, backed by the full power of 1,300+ tools. Results are organized by category -- regulatory, research, clinical, safety -- so you can drill into exactly the information you need without wading through noise.
Quickly verify drug interactions, check contraindications, and review FDA safety communications. ZonzNet replaces the need to manually cross-check multiple databases during patient consultations.
Accelerate literature reviews by searching PubMed, clinical trial registries, and institutional research databases simultaneously. Surface relevant studies faster and discover connections across disciplines.
Equip your teams with a single tool that consolidates drug information from the most trusted sources. Reduce time spent on information retrieval and improve decision-making accuracy across your organization.
Access 1,300+ healthcare tools and data sources from a single dashboard. No setup required.
Visit ZonzNet ↗